- Overview
- Amelogenesis
- Initiation
- Apposition
- Mineralization
- Clinical considerations

Figure 9.1: An illustration of the large number of and complicated interplay between morphogens involved in amelogenesis. Image credit: The sequential and reciprocal regulatory signaling between epithelium (red) and mesenchyme (blue) by Thesleff, I. and Tummers, M, Stembook.org, is licensed under CC BY 3.0
Overview
Even though dentin formation occurs before enamel formation, we start with enamel formation. This way, the chapter on dentin formation can remain close to pulp, which is important because they share the same lineage from neuro-mesenchyme. Enamel is produced by cells derived from the ectoderm. This leads to key molecular difference between the extra-cellular matrix of enamel versus that of dentin, cementum and bone tissue.
Another major difference we see is that enamel-producing cells are lost during tooth eruption, whereas the cells that produce dentin, cementum and pulp remain within a vital tooth. One major focus of studying embryology is to learn about how stem cells differentiate into adult cell types, in case we ever find the need to make adult cells de-differentiate and then re-differentiate into a cell type that needed. Re-creating ameloblasts would might not be terribly useful, because enamel is made from the surface of the crown, unless you could protect them with some sort of covering. But ameloblasts and odontoblasts undergo reciprocal signaling, meaning you can’t get odontoblasts without ameloblast signals, and you can’t get ameloblasts without odontoblasts, it takes a team effort.
In this chapter, we mention two or three morphogens (ones we’ve seen before), and one transcription factor. Hopefully Fig. 9.1 illustrates that this process is more complex.

Figure 9.2: Animated representation of odontoblast and ameloblast formation.
Initiation
This paragraph should be review. Amelogenesis starts at the bell stage of tooth development. At this stage, ectodermal cells invaginate to produce two layers of simple cuboidal cells called the Inner Enamel Epithelium (IEE) and Outer Enamel Epithelium (OEE). Between the IEE and OEE, more epithelial cells are scattered, namely the stratum intermedium and stellate reticulum cells. Collectively, these cells are known as the Enamel Organ.
Let us now add details to what we learned in the previous chapter. The enamel organ grows around a cluster of neuro-mesenchymal stem cells known as the dental papilla. Initially, a basement membrane separates the two. The first visible sign of differentiation occurs when the cuboidal IEE cells next to the dental papillae elongate, becoming more columnar in shape. Their nuclei move to the apical side of the cell, while organelles needed for secretion accumulate on the basal side. We therefore say the pre-ameloblasts undergo polarization. It is not known what triggers this change, a good first guess would be one of the different planar cell polarity morphogens. The ability to undergo mitosis halts for these cells at this time, meaning no new ameloblasts can form after the bell stage.
The disappearance of the basement membrane allows these cells to move more easily, due to the loss of hemi-desmosome attachments. The pre-ameloblasts secrete a number of short-range morphogens which induce neighboring neuro-mesenchymal cells to differentiate into odontoblasts and begin secretion of pre-dentin.
This puts pre-ameloblasts in direct contact with pre-dentin. Integrins on the pre-ameloblasts now contact a different form of collagen found in pre-dentin (type I), as opposed to the type in the basement membrane (type IV). In addition, newly formed odontoblasts secrete several morphogens, including BMPs. Collectively, these signals are the first signal in the differentiation of ameloblasts.
The second signal comes from the opposite direction, the stellate reticulum. One such signal produced by stellate reticulum cells includes a member of the Fibroblast Growth Factor (FGF) family.
This leads to the activation of transcription factors in the pre-ameloblasts, which up-regulate the expression of genes involved in the secretion of enamel. Transcription factors involved in amelogenesis include MSX-2, a homeobox gene, and RUNX2, a transcription factor which is also involved in osteoblast differentiation. Visible under the microscope, pre-ameloblasts change shape again, becoming shorter, and develop a bump on the basal side known as Tomes’ process. A summary of some molecules involved is in Table 9.1. The names are less important than the general order of events: from morphogen to receptor to transcription factor to protein expression.
| Morphogen examples | Receptors | Transcription factors | Genes up-regulated |
|---|---|---|---|
|
Collagen (dentin) BMP (Odontoblasts) |
Integrins BMP-R |
MSX-2 DLX1 RUNX2 |
Enamelin Amelogenin Ameloblastin Matrix Metalloproteinase |
|
FGF (Stellate Reticulum) |
FGF-R |
Table 9.1: a partial list of molecules involved in ameloblast differentiation

Figure 9.3:A thin section highlights enamel’s colorless state. Image credit: “18036” is in the Public Domain, CC0 / cropped
Apposition
After changing morphology, ameloblasts secrete enamel matrix from Tomes’ process. This ECM is similar to the ECM of bone tissue, with some key differences. Like bone matrix, enamel matrix is first secreted in a softer form, consisting mostly of proteins and water, and it mineralizes later. Enamel matrix contains CaPO4 crystals and organic components. Enamel, after it matures, has a higher mineral percentage, making it the hardest substance in the human body. Unlike bone tissue, however, enamel matrix has very little or no collagen. Instead, ameloblasts secrete similar proteins known as Amelogenins and Enamelins. These proteins act as nucleation centers to help induce calcium and phosphate to form crystals. The important fact to remember here is that collagen is a protein made by Fibroblasts (and other cells derived from mesenchyme), which come from the mesoderm or from neuro-mesenchymal cells. Ameloblasts lineage, on the other hand, is from the ectoderm. Presumably the connective tissue genes like collagen are methylated and packed away around histones, and are not available for transcription.
The shape of the enamel organ in the bell stage determines the shape of the crown. No new ameloblasts form because they lose the ability to undergo mitosis once they differentiate into pre-ameloblasts. Furthermore, no other stem cells can come into contact with both dentin and stellate reticulum once a thick layer of enamel pushes the two apart, so no new ameloblasts can form by differentiation, either. Because of how the pre-ameloblasts lined up in the bell stage, mostly pointing towards the oral cavity, enamel is thicker in the masticatory surfaces and thinner in the cervical areas.

Figure 9.4:Enamel is deposited appositionally, as seen by the Lines of Retzius. Image credit “Tooth of Paranthropus robustus SKX 21841 from Swartkrans” by Didier Descouens is licensed under CC BY 3.0 / red box added
Enamel is secreted by ameloblasts from Tomes’ process, laying down a new layer of pre-enamel over the older layers, pushing the ameloblasts further away from dentin. Ameloblasts, like many human cells, do not have a uniform speed. About every 4-12 days there is a change in the enamel deposition rate. As a result of deposition of enamel speeding up and slowing down, visible bands of lighter and darker enamel (less dense and denser) are visible when viewed in cross section. These visible bands are known as Lines of Retzius (or striae of Retzius, Fig. 9.4). One such band, the neonatal line, is particularly dark because the ameloblasts rested during the day we were born. After birth, the edges of the layers may be visible on the surface of teeth, especially the anterior teeth, in which case they are called perikymata. Over time, chemical and physical abrasion wears perikymata away. Counting the number of perikymata and multiplying by a number between 4 and 12 yields an estimate of how long it took the crown of that tooth to develop. This has no use in dental hygiene. It does represent a pattern unique to each individual, and thus may act like a fingerprint, with possible applications in forensics and biometric identification (a tooth print instead of a fingerprint).

Figure 9.5: Enamel rods are each produced by one small group of ameloblasts.
A layer of ameloblasts produce horizontal layers of enamel, new on top of old, but one ameloblast produces a single enamel rod (or prism) in a perpendicular direction. Because the rods are rounded, and not square, there are gaps between them. These gaps aren’t empty, they are filled with a slightly different form of enamel, called inter-rod enamel (while the enamel within rods is called rod enamel).

Figure 9.6: Hunter-Schreger bands are the enamel produced by a group of ameloblasts, and are lines that run perpendicular to the DEJ. Image credit: “18036” is in the Public Domain, CC0 / cropped
If you look closely to Fig 9.6, you might notice Hunter-Schreger bands (HSB), which are lines running perpendicular to the lines of Retzius. Each line represents the contributions of a small group of ameloblasts. If it’s a little hard to see, that could be because each enamel rod is microscopic. What you are looking for is a pattern similar to the fore edge of a textbook (all the pages as a group). Ameloblasts do not work in perfectly straight lines, the bands tend to curve slightly. Furthermore, different regions have different curve angles and patterns. This increases the tensile strength of enamel. If you have ever chopped wood, you know it is easy to do if you hit the axe right along the grain. Hit at an angle, and it can hurt you instead. The bending of Hunter-Schreger bands ensures there is no single grain along which a tooth could split. Cracks develop, but they tend to not extend the whole way through. The planar cell polarity signals that guide rod organization have been identified (Antagonists of Wnt morphogens).
As a fun side-note, or perhaps of relevance to those planning on pursuing a career as a veterinary dental hygienist at your local zoo, the HSB of hyenas occurs in a zig-zag pattern, increasing the tensile strength of their enamel to allow them to chew through bones. Dentin has curves (plural) which get specific names.
Maturation
Enamel is first secreted as a protein-rich gooey substance called pre-enamel. Two major protein components of enamel are the proteins amelogenin and ameloblastin. Ameloblasts also secrete a digestive enzyme. This enzyme, a matrix metalloproteinase, digests collagen and makes it easier for ameloblasts to migrate though the ground substance of overlying mesenchyme. This is necessary because as ameloblasts deposit pre-enamel from Tomes’ process, it pushes the cells outwards. If you recall, matrix metalloproteinases are also involved in the proper migration of tissues during formation of the palate. Matrix metalloproteinases serve a second role by processing enamel proteins made by the ameloblasts after they are secreted into the extracellular matrix. This helps to ensure there is a delay between secretion of pre-enamel and the mineralization of enamel. Without a delay, ameloblasts could become trapped by their own enamel, halting amelogenesis.
Once pre-enamel has been laid down and mineralizes, ameloblasts switch tasks and cause the enamel to mature. Some of the proteins are removed, replaced by fluids secreted by the ameloblasts, which then crystalize. We can therefore think of some of the protein secreted by ameloblasts as a scaffold that ensures enamel can be made quickly, at about 4 μM per day. Fluoride-based re-mineralization of tooth enamel is not measured using a small ruler, suggesting the rate of re-mineralization is too slow to see under the microscope. Our increased understanding of scaffold proteins has propelled researchers and companies to develop better tools for assisting in enamel re-mineralization, which mimic the ability of amelogenins and ameloblastin proteins to nucleate the formation of calcium hydroxyapatite crystals from a solution containing calcium and phosphate. These products do not need to contain proteins, for instance xylitol and certain glass compounds nucleate calcium hydroxyapatite crystals. Xylitol has the dual benefit of replacing simple sugars in foods, removing the substrates which harmful oral bacteria use for energy to secrete enamel-dissolving acids.
A number of different electrolytes and molecules are incorporated into enamel during the maturation stage. One important electrolyte is Fluoride (F–). Fluoride ions are negatively charged, and incorporate into hydroxyapatite crystals by replacing a small number of hydroxide (OH–) ions. This has important clinical implications. First, a small amount of Fluoride increases the strength of enamel. Secondly, Fluoride ions found in either saliva or incorporated into enamel neutralize acids released by bacteria, reducing enamel demineralization and preventing caries. Excessive Fluoride levels, however, affect the activity of ameloblasts during amelogenesis, reducing their removal of protein scaffolds. This may create a visual change in the form of white lesions on the surface of teeth, where the enamel has a higher protein and lower crystalline content, known as dental fluorosis. Topical application, from toothpaste or gels, allows Fluoride to incorporate into the surface layers of enamel, which has considerable benefits. But the greatest potential benefit is when Fluoride exposure occurs during the apposition and maturation stages of amelogenesis, early in embryonic development.
Clinical considerations

Figure 9.7: Acid etching demineralizes rod and interrod enamel at different rates, producing a rough surface suitable for bonding. Image credit: This work by Janice Carr, USCDCP is in the Public Domain, CC0
Surface etching
Because rod enamel is slightly different from interrod enamel, applying an acid to the crown surface wears the two at different rates—a process called surface etching. This creates an uneven surface, providing more surface area to create a stronger bond between the tooth and a surface sealant (think about how dermal papillae and rete pegs interdigit with one another for a stronger connection). Treatment with a weak acid may also remove debris at the surface, known as a smear layer, which otherwise interferes with bonding of an adhesive.
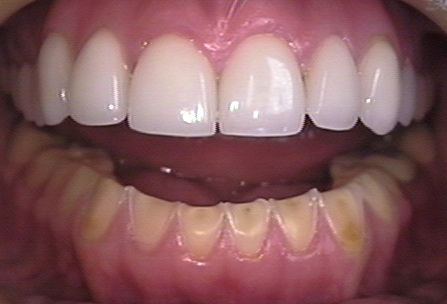
Figure 9.8: Oral manifestation of bulimia by Jeffrey Dorfman is licensed under CC BY 4.0
Erosion (or corrosion)
Prolonged exposure to a weakly acidic environment, or a shorter exposure to a highly acidic environment, can cause erosion of tooth enamel. Positively charged H+ ions of an acid react with negatively charges OH– and PO4– ions of calcium hydroxyapatite crystals, dissolving them. Bulimia and acid reflux bring highly acidic hydrochloric acid of the stomach in contact with teeth, causing erosion. Xerostomia, caused by the abuse of drugs such as methamphetamines, the use of certain medications, chemotherapy, or associated with old age, reduces the protective buffering ability of saliva to protect tooth enamel. Acidic foods or sugary foods may also contribute to enamel erosion. The simple sugars in many foods are nutrients oral bacteria metabolize and use to produce energy, which they then expend to create and secrete enamel-eroding acids.

Figure 9.9: Dental caries (red arrows) caused by impacted 3rd molars (green arrows). Image credit: “Impacted wisdom teeth caries” by Coronation Dental Specialty Group is licensed under CC BY-SA 3.0
Early detection of erosion (a dental caries, or tooth decay) can be done during a regular examination. Early signs of enamel erosion includes the appearance of a white lesion on the surface of enamel, which may be softer to the touch, and appear more radiolucent on a dental radiograph (Fig. 9.9). Treatment often involves removal of the carious tissue followed by replacement with a bio-compatible material.

Figure 9.10: Three common patterns of tooth abfraction, all along the dentin-enamel junction. Image credit: Own work, By Ebd2015 is licensed under CC BY 3.0
Abfraction
When non-carious tooth material is lost by force, it is called abfraction. Abfraction frequently occur along the dentin-enamel junction (DEJ). Chewing causes flexion forces at the cervical region of the tooth. Treatment of this type of lesion should involve reducing the stress that caused it in the first place, such as parafunctional habits or tooth occlusion. Because enamel is stronger than dentin, dentin is frequently lost in response to force transmitted through the enamel to the DEJ.
Developmental disturbances

Figure 9.11: An example of the pitting and white lesions caused by enamel hypoplasia. Image credit: Symmetrical enamel hypoplasia of grade I on permanent incisors in a CD patient by Maurizio Procaccini et al, Head & Face Medicine is licensed under CC BY 2.0
Enamel hypoplasia
Reduced enamel formation during embryonic development is called enamel hypoplasia. This occurs to primary and/or succedaneous teeth. Hypoplasia in an embryo is often triggered by health changes in the mother, such as a gastrointestinal disorder or a syphilis infection during pregnancy. If ameloblasts cannot acquire enough minerals during the time they are active, upon tooth eruption the surface enamel may contain white lesions, or exhibit pits and grooves. The enamel is more brittle and susceptible to erosion and abfraction. Furthermore, increased sensitivity to temperature and increased incidence of dental caries and periodontal disease are expected. Because amelogenesis is complete long before birth, there is currently no way to significantly assist in enamel production after tooth eruption occurs. Therefore, preventative treatment becomes much more important, and full-coverage crowns might be required.
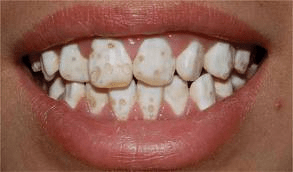
Figure 9.12: An example of a patient with amelogenesis imperfecta. Image credit: Amelogenesis imperfecta by 신연아 is licensed under CC BY 3.0
Amelogenesis imperfecta
Another cause of enamel hypoplasia is the genetic condition amelogenesis imperfecta. There are a number of different genes that, when mutated, cause this disease. For instance, mutations in the genes for amelogenin and enamelin lead to forms of amelogenesis imperfecta by reducing the ability of scaffolds to rapidly nucleate hydroxyapatite crystal formation. Another gene mutation that can be inherited is in a matrix metalloproteinase. Because amelogenesis imperfecta is caused by different mutations to a number of different genes, the symptoms occur on a spectrum, from mild to severe.

Figure 9.13: An enamel pearl. Image credit: Own work, By Wicketcity is licensed under CC BY-SA 4.0 / cropped
Enamel pearl
The IEE within HERS does not normally differentiate into ameloblasts because of the lack of morphogens from stellate reticulum. However, a small region of the IEE might mistakenly differentiate into ameloblasts along the root and produce a small amount of enamel over cementum, known as an enamel pearl. There is no harm in having pearls, other than they may be confused with a calculus during a scaling procedure. If the wrong type of dental scaler is used, the instrument tip may break off when it encounters the much harder enamel pearl. They can be identified by radiography, because enamel is more radiopaque than cementum. Enamel pearls generally occur on the furcations of the multi-rooted molars.