- Overview
- Dentin
- Induction
- Apposition
- Maturation
- Types of dentin
- Clinical considerations of dentinogenesis
- Pulp
- Anatomy
- Microscopic features
- Clinical considerations of pulp

Figure 10.1: Illustration of dentinal tubules. Order of appearance: odontoblasts, nerve endings, overlying gingiva and enamel.
Overview
Dentin and pulp are in this chapter together because of their shared lineage: they are derived from the neuro-mesenchyme of the dental papilla. Even though dentin resembles enamel superficially, by the end of this chapter you should see similarities between dentin and pulp. The two are often referred together as the dentin-pulp complex.
One key characteristic of dentin is that the extra-cellular matrix contains long tunnels that run through the whole layer, and these are filled with a little fluid and a cytoplasmic extension of a dentin-producing cell, the odontoblast. This have significance when it comes to dental hypersensitivity, as well as in the repair of dentin following damage.

Figure 10.2: Important regions during dentinogenesis. Legend: P = pre-dentin, D = mature dentin, arrows indicate cell bodies of odontoblasts found at the edge of the pulp. Image credit: Histological section of tooth by Doc. RNDr. Josef Reischig, CSc. is licensed under CC BY-SA 3.0.
Dentin formation
Dentinogenesis is the process of dentin formation (and should not be confused with the word odontogenesis). Dentinogenesis begins during the bell stage of tooth development. Odontoblasts (arrows in figure 228) are the cells that produce dentin, first by secreting proteins including collagen that act as a scaffold. Mineralization around the scaffold occurs later. The initial protein-rich material is pre-dentin (P in Fig. 10.2), after it mineralizes it is called dentin (D in Fig. 10.2).

Figure 10.3: Animated view of amelogenesis and dentinogenesis
Induction
Cells of the IEE differentiate into pre-ameloblasts and secrete morphogens, including a Bone Morphogen Protein. The outer layer of neuro-mesenchymal cells of the dental papilla receive these morphogens and differentiate into odontoblasts. This involves a morphological change, as the relatively shapeless neuro-mesenchymal stem cells line up with one another, forming what looks more like a simple cuboidal epithelium, complete with apical-to-basal polarity. This sort of polarization is unusual for a connective tissue, but odontoblasts are not derived from mesoderm, but neuro-mesenchyme. It is very common for neurons and glial cells to be polarized, and the odontoblasts do not lose this ability. Inside the odontoblasts, large amounts of endoplasmic reticulum and Golgi apparatus are forming in preparation for the secretion of large amounts of protein.

Figure 10.4: Polarization of odontoblasts, including cell-to-cell junctions that trigger activation of genes for proteins secreted into dentin. Legend: Green = cell-cell junctions help establish cell polarity by triggering intra-cellular signals. Yellow= secretion of collagen-filled vesicles to form pre-dentin (light blue layer) occurs at the apical surface of the cell body. Brown = secretion of enzyme-filled vesicles from the odontoblastic process triggers mineralization of pre-dentin into dentin (darker blue layer). Pink = pulp.
Apposition
Newly formed odontoblasts begin secreting proteins that are found within mature dentin and act as a scaffold during the mineralization of dentin. This protein-rich substance is pre-dentin (the lighter blue layer in Fig 230). It is mostly collagen plus a few other dentin-specific proteins. Pre-dentin later mineralizes, producing calcium hydroxyapatite crystals similar to enamel and bone tissue (darker blue layer in Fig 230). As layers of dentin mineralize, odontoblasts continue secreting new pre-dentin, pushing the layer of odontoblast cell bodies deeper into the jaw. Unlike ameloblasts, odontoblasts leave behind an odontoblastic process in the dentin they secrete. This arm-like extension contacts nearly every layer of dentin that odontoblast creates. Because dentin mineralizes around the odontoblastic process, a dentinal tubule runs through nearly the entire length of dentin. If you removed the odontoblasts, dentin would be perforated by millions of these tiny tubes.
Collagen and dentin-specific proteins are secreted by the side of the odontoblast cell body facing the enamel (yellow vesicles, Fig 230), which ensures that the cell body does not become trapped and instead touches a thin layer of pre-dentin. Proteoglycans and enzymes like Matrix Metalloproteinase are secreted a short distance up the odontoblastic process (less than 1mm, red vesicles in Fig 230) and trigger mineralization of dentin.

Figure 10.5: Inter-globular dentin (arrowhead). Image credits Figure 1a by Monaogna Vangala is licensed under CC BY-SA 3.0 / labels added
Dentin mineralizes in one of two ways, in globs or in lines. The first dentin that is produced mineralizes in globs because collagen is secreted in circular globs. Globular dentin mineralizes into mature dentin. Some regions do not mineralize, and are called inter-globular dentin. Low levels of phosphate during the first trimester can increase the amount of inter-globular dentin. After the first collagen fibers are secreted in globs, the fibers can be lengthened by odontoblasts. This dentin mineralizes more uniformly, one layer at a time, in a process called linear mineralization. Collagen fibers run parallel to each other in regions that mineralize linearly
The lines that should be clearly visible in the layer of dentin in Fig. 10.5 are dentinal tubules, the tubes in which the odontoblastic process are found. Notice that they do not run in a straight line. Odontoblasts, like ameloblasts, grow in a slightly curved direction, and this increases the strength of dentin. The large curves are called the primary curvature—if you zoomed out you would see a sinusoidal (back and forth) shape. This curvature is more pronounced in the crown than the root. If you zoom in on a single odontoblastic process, you might see areas where it curves back the opposite direction a little. That is called a secondary curvature. In contrast, the bending of enamel rods is called “wavy”, there are no names for bigger and smaller curves.

Figure 10.6: Imbrication lines of Von Ebner (in direction of red lines). Image credit “histological section of tooth” by Doc. RNDr. Josef Reischig, CSc. is licensed under CC BY-SA 3.0 / red lines added
Similar to the formation of enamel, odontoblasts undergo cyclic patterns of more and less rapid pre-dentin deposition. As a result, light and dark bands found in dentin are visible, called the Imbrication lines of Von Ebner (or the incremental lines of Von Ebner), which are comparable to the Lines of Retzius seen in enamel. Don’t ask how Retzius saw lines in enamel but missed the same pattern in dentin a few millimeters away, leaving that to Von Ebner to discover a hundred years later. People get credited for re-discovering things all the time, such as Christopher Columbus. These should both be renamed incremental lines in enamel and the incremental lines in dentin, anyway.
Particularly prominent imbrication lines can, furthermore, be called contour lines of Owen. This includes the neonatal line, similar to the one found in enamel. These represent big changes in nutrition that lead to changes in the density of dentin produced that day. You can read part of Owen’s treatise on the comparative anatomy of the tooth, someone scanned 30 pages if you are curious what it was like to learn about histology without pictures.
Maturation
Unlike enamel, dentin does not undergo significant changes after it mineralizes. As a result, some textbooks call the mineralization of pre-dentin into dentin as the “maturation” step, while others (including this one) have chosen to use the words apposition and maturation consistently. Why do ameloblasts take an extra step removing some scaffolding after mineralization occurs, and odontoblasts do not? One possibility is ameloblasts are epithelial cells, and epithelia generally secrete very little ECM, and are not very good at it. Odontoblasts differentiate from neuro-mesenchymal stem cells. It is likely during their epithelial-to-mesenchymal transition they unpacked genes that make them more efficient at creating ECM. A second possibility is that the increased percentage of minerals in mature enamel can only occur after protein scaffolding is removed, leaving behind a higher percentage of minerals. The reason is not as important as the illustration of another difference between enamel and dentin formation that can be traced to different cell lineages.

Figure 10.7: Dentinal tubules without odontoblastic processes. Image credit: Dentinal tubule occlusion of dentine discs after treatment by Peiyan Yuan is licensed under CC BY-SA 3.0 / cropped and animated labels added
In Fig. 10.7, dentinal tubules should be easily visible. In a living tooth, each would contain an odontoblastic process. Not all regions of dentin are the same, and there are several ways of classifying different types of dentin. One way is based on how close the dentin is to the dentinal tubules. The thin white area of dentin immediately surrounding each dentinal tubule is called peri-tubular dentin, while the rest is inter-tubular dentin.

Figure 10.8: Types of dentin by location. Image credit: “cross sections of teeth” by Gorak Tek-en is licensed under CC BY 3.0 / animation and text added
Another way to classify types of dentin is on its location relative to the pulp cavity. Mantle dentin forms first, and is a thin layer (15-30mm) closest to the enamel. Mantle dentin contains few dentinal tubules, they are filled in during the maturation stage. Mantle dentin is where globular mineralization occurs. The collagen fibers that remain in mantle dentin run perpendicular to the DEJ. The rest is circum-pulpal dentin, which mineralizes linearly, leaving dentinal tubules intact. Mantle and circum-pulpal dentin have slightly different levels of mineralization and protein content. The collagen fibers in circum-pulpal dentin run parallel to the DEJ.
![[add-text output image]](https://i0.wp.com/histology-and-embryology.com/wp-content/uploads/2020/08/add-text-output-image-1.gif?resize=135%2C240&ssl=1)
Figure 10.9: Types of dentin by time of production. Original image “cross sections of teeth” by Gorak Tek-en is licensed under CC BY 3.0 / animation and text added
Yet another way to classify dentin is based on when it is formed relative to the apical foramen. If this seems redundant to mantle versus circum-pulpal dentin, you are mostly right. Primary dentin is dentin formed before completion of the apical foramen, therefore it is formed prior to tooth eruption. Secondary dentin is formed after the completion of the apical foramen (after tooth eruption). Unlike mantle versus circumpulpal dentin, there is no major histological difference between primary and secondary dentin.
Much later, a tooth may suffer damage. Odontoblasts in the pulp produce tertiary dentin and repair the damage. This may also be called reactionary dentin. Formation of this form of dentin involves secretion of matrix metalloproteinases from dentinal tubules. These enzymes are also used during the formation of dentin in embryogenesis, so this this is another example of wound repair recapitulating embryonic development. During embryogenesis, however, dentin is formed appositionally. Tertiary dentin mineralization occurs from a dentinal tubule, causing the tubules to become occluded. Therefore, fewer dentinal tubules are found in reactionary dentin. Furthermore, it is the parallel dentin tubules that give primary and secondary dentin their yellow-ish hue. With reduced or no tubules, reactionary dentin becomes more translucent (see Fig 238).

Figure 10.10: Osteodentin (arrow) inside of the pulp cavity. Image credits “Tooth, Pulp – Osteodentin in a male F344/N rat from a chronic study” by Cora MC, Travlos GS. 2014. , National Toxicology Program is in the Public Domain, CC0
If a large enough injury occurs as to expose the pulp chamber, possibly destroying odontoblasts, a much more robust response is needed. Mesenchymal stem cells in the pulp are induced by morphogens to differentiate into new odontoblasts and form a type of tertiary dentin called reparative dentin. This form of dentin does not form the same way dentin does during development, which began at the dentin-enamel junction and moved deeper. Instead, to form reparative dentin, odontoblasts and fibroblasts migrate throughout the injured area (after a hematoma forms) and secrete proteins and electrolytes, trapping many of the cell within. No dentinal tubules are created when odontoblasts secrete dentin in all directions. For this reason, reparative dentin is sometimes referred to as osteodentin, because of its resemblance to bone tissue more than tubular dentin.

Figure 10.11: Sclerotic dentin (S). Image credits “Stereomicroscope image (5X) of tooth with measurement of Sclerotic Dentin Area (S) and Length of Sclerotic Dentin (SL)” by Selvamani M, et al, Journal of International Oral Health is licensed under CC BY-SA 4.0 / cropped
Lastly, if there are more fibroblasts than osteoblasts repairing the damage, the end result is more of a calcified scar than dentin, some textbooks call this sclerotic dentin. Sclerotic dentin is mostly collagen and calcium hydroxyapatite crystals.
| Type of dentin | Location | Features | |
|---|---|---|---|
|
Peri-tubular |
Walls of tubules |
||
|
Inter-tubular |
Between walls |
||
| Mantle | Thin border next to DEJ |
No tubules Collagen perpendicular to DEJ |
|
| Circum-pulpal | Rest of tooth |
Tubules Collagen parallel to DEJ |
|
|
Primary |
Formed before apical foramen |
Made by original odontoblasts Contains tubules |
|
|
Secondary |
Formed after apical foramen |
||
|
Tertiary |
Reactionary |
Formed after small injury |
Made by new odontoblasts Fewer tubules |
|
Reparative and sclerotic |
Formed after significant injury |
Made by new odontoblasts and fibroblasts Cell trapped in calcified tissue (osteodentin) |
|
Table 10.1: Summary of the types of dentin.
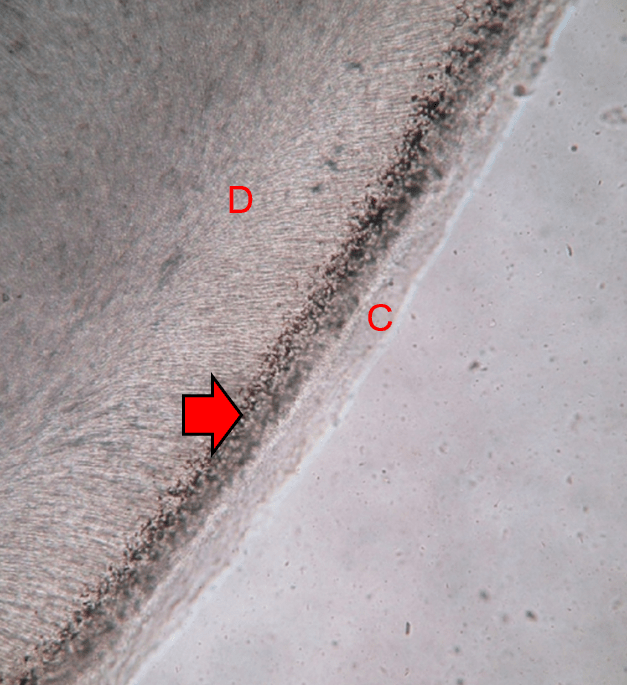
Figure 10.12: Tome’s granular layer (arrow) found in root dentin (D), just deep to cementum (C). image credit “Tomes granular layer “ by Shaik Mohamed Shamsudeen is licensed under CC BY-SA 3.0 / letters and arrow added
Root dentin
Roots do not contain mantle dentin, but they do contain a superficial layer of dentin that is visually distinct from deeper regions. Close to the border with cementum, spots are visible in a band of dentin known as Tomes’ Granular Layer. It has no known clinical significance; it is simply a grainy area. It is useful for orienting yourself when looking at histological sections of teeth. Those grain are not the nucleuses of cells, nor are they inter-globular dentin. Older data suggested the grains represent loops of dentinal tubules, but re-analysis using more advanced microscopes suggests the grains are large bundles of collagen fibers.
Clinical considerations of dentinogenesis

Figure 10.13: Electron micrograph of the interface between dentin and a mineral aggregate (MTA) used in the a root canal therapy to boost replacement of resorbed root dentin. Image credit: “Scanning electron microscopic images that are representative of the root canal dentin and mineral trioxide aggregate (MTA) interface” by Yoo, Yeon Gee et al is licensed under CC BY 3.0
Mineral aggregates
If there is a large amount of damage to dentin, the production of tertiary dentin may be too slow. For cases such as treating root resorption or for root end filling during endodontic therapy, artificial ECM can be used. Unlike some of the artificial tissues discussed in gingival healing, hard substances generally not make good scaffolds, as the density of the matrix inhibits migration of stem cells. The goal of using mineral aggregates is to provide the necessary materials required by odontoblasts without inhibiting their movement during dentinogenesis. One such compound, Mineral Trioxide Aggregate (MTA), was developed in California by Dr. Mahmoud Torabinejad. MTA contains a purified version of Portland cement plus calcium-containing minerals. The addition of mineral aggregates speeds up the formation of tertiary dentin. MTA slowly releases calcium hydroxide, which provides a raw material for mineralization of tertiary dentin, as well as attracting phosphate from the blood or ECF.
Author’s note: the authors currently live in or near Portland, Oregon, but Portland cement was invented in Portland, England. Do not apply Portland cement from your hardware store to teeth, it is caustic and may contain arsenic or other heavy metals. The Portland cement used in dentistry is purified.

Figure 10.14: The appearance of dentin should be glistening and moist. Image credit: “Dentin appearance” by Pinkmanggis is licensed under CC BY-SA 4.0
In one type of dental restoration (a tooth filling), a wet resin is bonded (glued) to dentin and hardened using a special light. To bond resin to dentin, one should take into account the chemical structure of dentin. The hydroxyapatite crystals and collagen of the ECM of dentin contain some water molecules, giving dentin a moist appearance. Many bonding agents do not adhere well to a wet surface, and require dentin to be dried first. This is usually done with a volatile solvent such as ethanol. Mineral acids may be used to remove an amount of the mineral ECM of dentin, leaving behind the more porous collagen framework which can improve bonding. This is similar to acid-etching of enamel, but there are no rods and inter-rods to take advantage of in dentin.
There is interest in the research community for developing bonding agents that adhere to wet surfaces (all living human tissues are wet). One promising area is the study of mollusk mucus. Mucus is secreted from the body, but it is similar to ground substance in human connective tissues, especially mesenchyme. Mucus secreted by mollusks like limpets adhere to wet surfaces very tightly, and has the potential to stimulate tertiary dentin formation (link to pdf download).
Morphogens

Figure 10.15: Wnt morphogens induce the differentiation of different cell types in different environments.
The formation of secondary and tertiary dentin requires morphogens to induce the differentiation of mesenchymal stem cells into new odontoblasts. As we learn more about these morphogens, it opens up the possibility to administer these morphogens and speed up natural healing processes, potentially reducing the need for inorganic cements or caps. One such morphogen is a Wnt (the W stands for wingless, because when the right one is mutated in flies it prevents the induction of wing placodes.) In the brain, Wnts induce the differentiation of neural crest cells into specific types of neurons and glia. But when neural crest cells migrate to the face and become the pharyngeal arches, the same Wnt induces them to differentiate into odontoblasts. The difference arises because of a second morphogen. This second signal comes from the ECM, and ECM is very different in the developing brain than it is in developing neuro-mesenchyme of the pharyngeal arches. The significance of this is that a drug used to treat Alzheimer’s Disease (AD), Tideglusib, is used off-label in dentistry to boost tooth repair. We suspect it is very rare to randomly test psychoactive drugs for their use in dentistry. Prior knowledge of shared lineages, however, makes new avenues of treatment more obvious.

Figure 10.16: Erosion of enamel and dentin. Image credit: Own work By Klaus Limpert, Scuba-limp is licensed under CC BY-SA 3.0
Attrition and erosion
Because dentin has a lower mineral content than enamel, it is lost more quickly than enamel. If mantle dentin is lost, this exposes dentinal tubules in the underlying circum-pulpal dentin. This increasing the amount of surface area for which acids can act upon, which speeds up dentin erosion further. Surface exposure of dentinal tubules also leads to increased sensitivity of the teeth (see below).

Figure 10.17: Caries in enamel versus dentin. Image credit Tooth By ADuran is licensed CC BY-SA 3.0
Dentin caries
Once a caries spreads through enamel, when it reaches the DEJ it spreads at an increased rate. The triangle pattern of enamel loss is quickly repeated in a new triangle of a dentin caries. It is possible, but not agreed upon, that a small enough enamel caries might spread to the DEJ without causing visible changes, and upon reaching the DEJ the rate of tooth loss accelerates. Whatever the exact cause, a caries can start below enamel and spread through dentin. This hidden caries is obviously harder to detect than one at the surface of a tooth.

Figure 10.18: Tooth hypersensitivity due to exposed dentinal tubules.
Tooth hypersensitivity
The loss of enamel or cementum covering dentin may expose dentinal tubules. In the crown, the few millimeters of mantle dentin at the outer surface contain few open dentinal tubules, but as that dentin is lost many more tubules in the circum-pulpal layer are exposed. These tubules extend all the way to the root pulp, where many nerve endings are located. Changes in the environment of the oral cavity, such as temperature, pH, alcohol level or osmolarity changes can now affect the temperature, pH, alcohol level or osmolarity of the pulp ECF. Odontoblastic processes do to entirely fill up the dentinal tubule, there is a small amount of ECF between the process and peritubular dentin whose chemical composition can change. To ECF can be detected by nerve endings that relay painful stimuli to the brain. This is called tooth hypersensitivity, or more accurately dentin hypersensitivity (DH). Treatments for DH include special toothpastes, mouthwashes or chewing gums that deposit minerals into exposed dentinal tubules and occlude them. Such products likely contain salts and fluoride. Alternately, varnish mineral aggregates such as Portland cement might be applied to the affected area.
Conversely, with age, odontoblasts add layers of peritubular dentin inside dentinal tubules, causing the tubules to become narrower. As this happens, teeth become less sensitive. This may not sound so bad until you take into consideration the role that proprioception has in preventing excessive occlusal forces. Cranial nerve V is one of the larger cranial nerve, it transmits a lot of important sensory information to the brain from the teeth. With diminished sensitivity comes an increased risk of occlusal trauma.

Figure 10.19: The importance of proper dental hygiene procedures. Image credit “Dental details” by Senior Airman Kristi Emler, US Air Force is in the Public Domain, CC0
Dentin may become exposed due to loss of cementum or enamel, or if the edge of enamel does not meet/overlap cementum. Furthermore, gingival recession exposes the thin layer of cementum to environments it is not designed to handle, making dentin exposure along the neck of the tooth the most likely area for DH. Improper technique on the part of dental hygienists or dentists may inadvertently remove protective layers of cementum as well. There is no reason to believe this is the case in figure 10.19, government agencies are simply a great place to find copyright-free images.

Figure 10.20: Dentin resorption (arrow). Image credit “An irregular radiolucency in the coronal third to middle third of the root” by Fang-Chi Li is licensed under CC BY-SA 3.0
Dentin resorption
During the shedding or primary teeth, dentin resorption—the loss of dentin due to cells re-absorbing minerals– assists in the loss of attachment between the tooth root and alveolar bone. This is mediated by odontoclasts. Compared to their relative the osteoclast, which have baseline activity throughout life maintaining bone tissue as part of a remodeling unit. Odontoclasts are not always present, their differentiation from mesenchymal stem cells as well as their activity is regulated by different morphogens.
Dentin resorption can be improperly triggered in cats, causing feline odontoclastic resorptive lesions. There is a significant amount known about this process in cats. In humans, dentin resorption occurring at any time other than the shedding of primary teeth is idiopathic (of unknown cause). We do understand that the dentinal tubules found throughout dentin create a large amount of surface area for odontoclasts to adhere to and trigger demineralizion. This is similar to what we observe in spongy bone: spongy bone is lost at a faster rate than compact bone because it has a higher amount of surface area, thus symptoms of osteoporosis appear first in bones which higher amounts of spongy bone, like the mandible. Dentin resorption can be triggered from the pulp-cavity side and be referred to as internal resorption. Conversely, external resorption occurs from the DEJ or CEJ side.
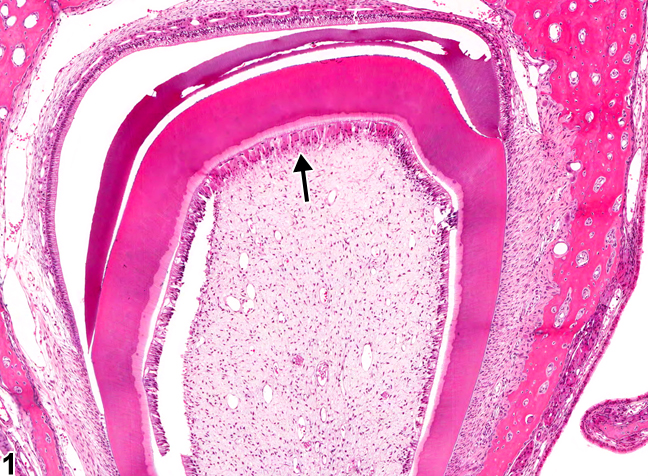
Figure 10.21: Histology of pulp. Arrow indicates odontoblastic layer. Image credit “Tooth, Odontoblast necrosis” by Cesta MF, Herbert RA, Brix A, Malarkey DE, Sills RC (Eds.), National Toxicology Program Nonneoplastic Lesion Atlas is in the Public Domain, CC0
Pulp overview
The dentin and pulp both develop from neuro-mesenchymal cells of the dental papilla. Odontoblasts make a more specialized tissue, containing just one terminally-differentiated cell type. The rest of the neuro-mesenchymal cells make a more generic tissue, areolar connective tissue, which contains numerous cell types, including adult stem cells. It is here that blood vessels, nerve fibers and lymphatic vessels have space and support to grow.
Histology of pulp

Figure 10.22: Coronal and radicular pulp. Image credit “Longituinal tooth 5” by Natdental is licensed under CC BY-SA 4.0
Pulp can be divided into coronal pulp and radicular pulp. The coronal pulp is in the crown of the tooth and contains smaller pulp horns beneath the cusps. Radicular pulp is in the roots, and may extend into accessory canals. Accessory canals connect the pulp to connective tissue external to the tooth, traveling laterally rather than out the apical foramen. Accessory canals form when HERS runs into a blood vessel and is forced to grow around it. Otherwise, radicular pulp terminates at the apical foramen.

Figure 10.23: Layers of the pulp. Legend:. 1) odontobalst layer 2) cell-free layer 3)cell-rich layer 4)pulp core. image credit “Pulp Histology © School of Dentistry, University of Dundee” by Dododopamine is licensed under CC BY-SA 4.0
Pulp layers
Pulp is a single tissue, but there are four layers that appear distinct from one another under the microscope. The first is the odontoblast layer, which is closest to the dentin. Odontoblasts initially form a single layer of cells along the broad region of the DEJ. But as they add dentin the pulp cavity becomes smaller, and the cell bodies crowd together, ultimately forming a thicker layer of cell bodies at the periphery of the narrower pulp cavity wall. Deep to the odontoblasts is what is called the cell-free zone. This zone contains cells, but they are not visible under a traditional H&E stain. Deep to that is a cell-rich zone, composed primarily of fibroblasts, mesenchymal stem cells, white blood cells and other connective tissue cells. Lastly is the pulp core, which contains the same types of cells, but more ground substance spaces them apart. The pulp core is where most of the blood and lymphatic vessels are located.
Clinical considerations of pulp

Figure 10.24: Gumboil caused by an underlying periapical abscess. Image credit “abcès parulique” by Damdent is licensed under CC BY-SA 3.0
Periapical abscess
If damage to dentin exposes the pulp to oral bacteria, an infection of the pulp may occur. This usually triggers inflammation of pulp tissue, known as pulpitis. Death of pulp tissue can lead to accumulation of pus within the pulp chamber, which is called a periapical abscess. The release of inflammatory molecules resulting from tissue damage can lead to swelling of the overlying gingival tissue, often referred to as a gumboil. A gumboil may also form due to an infection of periodontal tissues, in which case it would not be a periapical abscess, it would be a periodontal abscess. It is possible to remove necrotic pulp surgically. If some healthy pulp remains, regeneration of the pulp can occur. This is because of the high degree of vascularization of areolar connective tissue and the high mitotic potential of mesenchymal stem cells.

Figure 10.25: Endodontic therapy (root canal). Image credit Tooth #30 By DRosenbach at English Wikipedia is in the Public Domain CC0
Endodontic therapy
A periapical abscess likely requires the removal of the infected pulp tissue and replacement by a material that is similar in density and elasticity, a procedure known as endodontic therapy. The preferred material used is gutta-percha, a naturally-occurring latex polymer that is the sap of trees in Malaysia. It was once widely used as an insulator for electronics, but has been replaced by synthetic polymers, except in endodontic therapy. It is radiopaque because of an additive, barium sulfate. Otherwise, latex does not show on radiographs, which would make it difficult to confirm gutta percha had completely filled the pulp cavity.

Figure 10.26: Discoloration of a non-vital tooth after endodontic therapy. Image credit “Discolored maxillary left central incisor tooth” by Anandkumar Patil is licensed under CC BY-SA 4.0
Removal of living pulp tissue removes all cells from the tooth, including odontoblasts. This tooth is referred to as non-vital. A non-vital tooth cannot make repairs to dentin, which leads to increased brittleness and the accumulation of stains over time. Stain molecules may invade from the enamel side, or molecules from pulp necrosis may invade dentin by traveling up open dentinal tubules.

Figure 10.27: Pulp vitality testing. Image credit Own work By Coronation Dental Specialty Group is licensed CC BY-SA 3.0
Pulp vitality testing
Painful stimuli at the surface of the teeth are detected by nerve endings located within the pulp. Pulp vitality testing takes advantage of this to estimate the health of the tooth pulp. An electrical stimulus is applied at the surface of teeth followed by self-reporting of the discomfort level experienced by the patient (ideally, medical tests should not be painful). Loss of nerve endings reduces tooth sensitivity, and indicates pulp tissue has undergone fibrosis or necrosis. However, a reduction in the diameter of dentinal tubules also reduces the ability of electrical signals at the surface to be detected within the pulp. Conversely, loss of enamel and exposure of dentinal tubules may increase sensitivity without any changes to pulp vitality.

Figure 10.28: A pulse oximeter (A) and dental adapter (B used instead of pulp vitality testing). Image credit: “Pulse oximeter and sensor with specially manufactured adapter” by Lorena Ferreira Lima, et al is licensed under CC BY 4.0
Because different patients report pain differently, and because factors besides the health of the pulp affect the degree of discomfort reported, false-positive and false-negative results are possible with pulp vitality testing. Other less-invasive tests exist, including pulse oximetry and laser doppler flowmetry. These measure the vascular supply to each tooth, which correlates with the vitality of the tooth. Pulse oximetry takes advantage of the color change hemoglobin undergoes as it picks up oxygen. Laser doppler flowmetry takes advantage of the doppler shift waves exhibit when they bounces off moving objects (such as flowing red blood cells) but not stationary ones– similar to the way the sound of a motorcycle changes pitch when it is rapidly travelling toward you or away from you.
Age-related changes
Because a vital tooth contains a layer of living odontoblasts, dentin becomes gradually thicker with age. This is most noticeable in the fine pulp horns, as a result, the pulp horns recede with age. Furthermore, as with many cells, the mitotic ability of mesenchymal stem cells within the pulp decreases with age. As a result, older pulp tends to contain more scar tissue (collagen fibers) and has decreased regenerative capability and diminished sensitivity.