- 4 main tissue types (by adult appearance)
- epithelia
- connective tissue
- muscle
- nervous
- 3 main tissue types (by embryonic lineage)
- ectoderm
- mesoderm
- endoderm
Figure 2.1: Histology of areolar CT, highlighting the cells, collagen fibers, elastic fibers and ground substance.
A short history of histology
The aim of this chapter is to review histology covered in your Anatomy & Physiology classes– but only the tissues that appear in the head and neck, and only the parts that are relevant to this course. This is by no means a comprehensive review. If you need an extra review on basic histology, try these links:
- An interactive histology flowchart by Laird C. Sheldahl, Ph.D.:
- The Internet Pathology Laboratory for Medical Education
- at The University of Utah, Eccles Health Sciences Library.
- Histologyguide.com
- by T. Clark Brelje, Ph.D. and Robert L. Sorenson, University of Minnesota.
Histology is the study of what tissues look like under a microscope. A tissue is a group of cells, all of the same major type, working together to perform a function. Because most cells are transparent, microscopists use of a number of different stains to highlight different parts of cells. Fig 2.1 is an image of a very common staining technique using the dyes Hematoxylin and Eosin (H&E). These two stains turn molecules with negative charges blue, and positive charges pink. DNA and RNA take up both colors, which makes nucleuses purple, while most proteins turn pink. Unfortunately, most cells are full of proteins and their cytoplasm turns pink, but the extra-cellular matrix is also full of protein, which turns pink as well. We are therefore often trying to make sense of a sea of pinks with purple spots. On the plus side, if you purchase a histology coloring book, you only need two crayons.

Figure 2.2: Example of a confocal microscope image. Image credit: : Fluorescent cells by NIH image is in the Public Domain CC0
Imaging
Most of the images you see in histology textbooks use stains such as H&E stains– this type of technology dates back to the 1800s. Microscope technology has improved since then, such as the image in Fig. 2.2, which comes from a confocal microscope. The confocal microscope uses expensive lasers to generate very beautiful images, but relies on stains which are theoretically similar to H&E. Sadly, most textbooks still use the old-fashioned (uglier) images. Since you should be using neither the newer nor the older microscopes in your line of work, we have illustrated most of the histology images in this book. This allows us to focus on the concepts, and spend less time developing the skills necessary to interpret pictures of pink blobs with purple spots.
We presume that in the Anatomy and Physiology classes you took to get into this program, you learned to spot the differences between different types of tissues. We review those differences in this chapter. We ask you to keep an eye out for similarities between different tissues (aside from they all look like pinkish blobs with purple spots). If you jump ahead and read the embryology chapters, you’ll have a better idea of what to look for, but for now just keep that thought in the back of your head as we cover the major tissues of the human body.
Enough embryology to start learning about histology


Figure 2.3: One possible way to group cartoon faces, based on a few physical characteristics.
Classification: the old way, and a better way.
Histology categorizes tissues based on what the cells look like in adulthood. If we determined people’s families based off what they look like as adults, we might do an OK job of it, or we might make mistakes. For instance, we might lump the cartoon faces in Fig. 2.3 into two families based primarily on hair and eye color. That is how the 4 major tissues types we are going to review are categorized. The major problem with this sort of classification system is it is hard to tell what physical traits are due to nature (genetics, such as eye color) versus nurture (environmental factors, such as hair dye).

Figure 2.4: Another way to categorize cartoon faces, based off of lineage data.
When we learn about embryology, however, we see we can do better. We can categorize tissues using cell lineage instead, tracing who the adult cells are related to by following which cell they came from during embryogenesis. Early embryologists stained individual cells and followed them through cell division after cell division to see what they became. Others, such as Hans Spemann, moved cells from one place to another and asked whether they still turned into what was expected, based on their new location. When transplanting just a few cells lead to embryos with two complete heads, Spemann concluded that was unexpected. To get a better idea how that happens, science had to wait for the discovery of DNA-storing genetic information for an organism. Then, developmental biologists like Christiane Nüsslein-Volhard were able to determine the mechanisms by which a single embryonic cell became different types of cells in an adult organism–her organism of choice was flies, and for that work she won the Nobel Prize. Unlike earlier histologists and embryologists who named everything after themselves, we get fun names like Bicoid, Dickkopf, Frizzled and Sonic Hedgehog. These scientists followed the lineages of cells, watched what they become, and determined how the cells make decisions as to when and what to become.
Rather than use the appearance of our cartoon people, if we knew their lineage (you might also see the fancier term ontogeny), we can classify them into more accurate families, with one sub-family (Fig. 2.4). When you see the word lineage, think family line, where differentiated cells are the youngest generation, and stem cells are their parents and grandparents. Sadly, this is not how histology is taught, so we’re going to look at what adult cells look like and classify them based on physical characteristics. When we do, look to see whether two tissues have distinct borders (likely different lineages) or blended borders (often the same lineage).
| The 4 main tissue types |
|---|
|
Epithelial |
|
Connective Tissue |
|
Muscle |
|
Nervous |
Table 2.1: the 4 main tissue types

Figure 2.5: Example of an epithelium (with connective tissue below).
Epithelia
Location of epithelia
An epithelium is composed of epithelial cells, usually connected to one another in a sheet. There is very little ECM in an epithelium, mostly cells. The cells are anchored together by desmosomes, which holds them in a sheet. Tight junctions ensure that the things that travel from one side of the epithelium to the other pass through the epithelial cells, and therefore must either be lipid-soluble, or be recognized by transport proteins on the apical and basolateral plasma membranes. An epithelium therefore has apical-to-basolateral polarity. The apical surface faces the outside of the body, and the basolateral inward, and polarity means the two sides are different. The outside can mean the outer surface of the skin, or an inner surfaces, such as lining the oral cavity, stomach, and bladder. Every surface of the human body is an epithelium. Therefore, under the microscope, if you see an empty space, an epithelium borders that space. The inner lining of a sweat duct is an epithelium. The inner lining of a blood vessel is an epithelium. All other tissues are between that epithelium and the epithelium on the outer surface of our body. Even enamel is made by an epithelium, so if anyone tells you enamel is a connective tissue because it is looks similar to bone tissue, they haven’t studied embryology, and need to be educated. Use words like ontogeny.

Figure 2.6: Stem cells in the basal layer of a stratified epithelium.
Epithelia are thin
Epithelial tissue is good at healing because it contains numerous stem cells capable of undergoing mitosis (you can tell by the presence of visible chromosomes, rather than chromatin). For thicker epithelia, stem cells reside in the deepest layer. Epithelia cannot be terribly thick, however, because they are avascular (without their own blood vessels). Nutrients for an epithelium must diffuse from underlying connective tissue, which is why in Fig 2.6, the epithelial cells on the apical side lack nuclei. They are dead, but still connected to the rest of the epithelium. The down-side to being good at mitosis is it means those cells are a few steps closer to becoming cancerous. Many people regularly expose themselves to high doses of carcinogens found in alcoholic beverages and tobacco (chewing tobacco in particular), which leads to oral cancers.
In addition to membranes, glands are made of epithelial cells. Exocrine glands, such as the salivary glands, contain a single layer of epithelial cells, rolled up into tubes called ducts. These ducts bring exocrine secretions to a surface of the body. Endocrine glands, however, are different. They are not arranged in a sheet, but are amorphous clusters of epithelial cells. Endocrine glands aren’t avascular, either. Blood vessels grow right into the epithelia of an endocrine gland. This is important because endocrine glands secrete hormones directly into the blood.
| Number of layers | Shape of cells |
|---|---|
|
Simple |
Squamous |
|
Stratified |
Cuboidal |
|
Columnar |
Table 2.2: The basic categorization of most epithelia.
Epithelia are mostly classified based on two criteria. With a few exceptions, epithelia have a name from the first column and a name from the second column of Table 2.2. Simple epithelia have just one layer of cells, while stratified epithelia have more than one. Squamous means the cells are flat, like fried eggs. Cuboidal means square-ish in appearance (cells are 3D, but look 2D under the microscope). Columnar means the cells are tall (like columns).

Figure 2.7: Simple squamous epithelia (endothelia) forming the walls of capillaries.
Simple squamous epithelia
If you look at the lining of a capillary, you might be able to see the simple squamous epithelium that lines the inner surface. These cells, called endothelial cells, are functionally really cool cells, even if they are hard to see. Normally, endothelial cells keep blood inside of a capillary, but they can become damaged, such as during probing, flossing or coughing.

Figure 2.8: Simple cuboidal epithelia, histology and illustration. Legend: cuboidal epithelial cells (pink). underlying connective tissue (light blue) and lumen of tubes (white).
Simple cuboidal epithelia
Simple cuboidal epithelia line most exocrine gland ducts, such as sweat and salivary glands. In Fig 2.8, you should see two ducts coming out towards the camera, and one duct running side-to-side. The lumen of each duct is lined by a simple cuboidal epithelium. The cuboidal cells in the epithelium are connected to their neighbors on the sides by anchoring junctions. Where two ducts get close together, it is two simple cuboidal epithelia, not one stratified one, there are two separate layers of epithelia, with a little ground substance between them. Stratified cuboidal epithelia exist, such as in parts of the parotid salivary gland, but they are rare enough that we can skip them. The capillaries in Fig. 2.7 are lined by flat cells to allow for diffusion of nutrients out of the capillary and waste products in, while salivary ducts in Fig. 2.8 have thicker cells to keep saliva from leaking out and dissolving connective tissues of the face.
Figure 2.9: Histology of a simple columnar epithelium (highly folded), with illustrations highlighting the columnar cells (pink), goblet cells (blue) and the underlying connective tissue (lighter blue).
Simple columnar epithelia
Simple columnar epithelia are not found in the adult oral cavity, but in an embryo the columnar Inner Enamel Epithelium differentiates into enamel-producing ameloblasts during embryogenesis. The cells’ shape isn’t important other than it helps histologists to identify these cells in a developing tooth.

Figure 2.10: Lateral view of bed, showing a cat between the basolateral surface of the comforter and the apical surface of the thin bed sheet. Image credit: “photo” by unknown is in the Public Domain, CC0
If you are looking at the illustration in Fig 2.9, you may wonder why there are fewer cartoon nucleuses than visible in the histology picture. Did the illustrator mistakenly give you a picture of a stratified columnar epithelium and try to pass it off as simple? No. When we say an epithelium is simple, it means one cell thick from apical-to-basolateral. But when we cut a tissue sample to view under the microscope, the slice may be several cells thick from side-to-side. And when we lay the tissue sample on its side, the cells that were running side-to-side are now sitting on top of one another. We have illustrated what a simple epithelium should look like if we had the ideal one-cell-thick slice of tissue. This might be a good time to bring up perspective. Epithelia form flat sheets, and in Fig. 2.9 we are looking at a (folded) sheet in cross section. Imagine slicing through your bed and looking at it from the side view. You could make out the individual, single layers of your bed sheet and comforter. Furthermore, you might note that while each is a single layer, the bed sheet is thin and the comforter is thicker. You could identify the basolateral surface of your bed sheet (the side that touches you when you sleep) and the apical surface (the side close to the comforter). In Fig. 2.10, if there was a second cat on top of the first cat, we could say the layers of cats is stratified. But if the bed sheet was covered in a single layer of cats, side-to-side and front-to-back, the layer of cats would be simple. Under the microscope, we’re doing the same thing, but unfortunately cells are semi-transparent, so it gets messy. When we call this a simple epithelium in Fig. 2.9, that means there is only one pink cell between the white space (here, the lumen of the intestines) and the very light-blue space (the connective tissue, where nutrients can be absorbed into blood vessels). Those cells are thicker than cells we looked at in Fig. 2.8, but there are no columns on top of other columns.

Figure 2.11: Histology and cartoon of a stratified squamous epithelium (and a little connective tissue below)
Stratified squamous epithelia
Stratified squamous epithelia can be found in the outer lining of the skin, and the inner lining of the oral mucosa. While we have different names for the skin and the oral mucosa, if we were to name them based off of embryology, rather than superficial appearance, they would both contain a stratified squamous epithelium. Chapter 3 covers in more detail the epidermis of the skin, which is a keratinized stratified squamous epithelium. Keratin is a tough, water-resistant protein made by the main type of epithelial cell in this tissue, the keratinocyte. The stratified squamous epithelium changes at the vermilion border to less-keratinized, and the stratified squamous epithelium of the oral mucosa is either less or not keratinized. Based on that information, should we classify the vermilion zone as an extension of the labial mucosa, or as part of the skin and lips? Or is there a line in the vermilion zone that divides into a skin half and an oral mucosa half? It’s times like this we could use the advice of an embryologist to inform us we shouldn’t always look for lines in adult tissues, nor define borders between the skin, the vermilion and the oral mucosa based on color. If you change careers and go into proctology, use this same information down there.
Stratified cuboidal, stratified columnar and transitional epithelium
These tissues are not appearing in this textbook.
Figure 2.12: Pseudostratified epithelium, histology and cartoon. Order of appearance: epithelial cells, goblet cells, underlying connective tissue.
Pseudo-stratified epithelia
Ciliated pseudo-stratified columnar epithelium, or as many people say, pseudostratified epithelium, can be found in upper portions of the respiratory tract, such as lining the nasal cavity and the para-nasal sinuses. This tissue definitely deserves a different name from the stratified squamous epithelia of the skin, labia and oral mucosa. However, it probably didn’t need to break the naming rules outlined in Table 2.2. A pseudostratified epithelium has more than one layer, but it is hard to count how many. As a result, someone named this tissue pseudo-stratified.
The big fat blue cells in Fig. 2.12, which don’t have cilia, are goblet cells. These cells produce mucus. Goblet cells synthesize mucous proteins within their rER and secrete them, and when they attract water, they become mucus. This is very similar to ground substance, however mucus is secreted out of the body, ground substance is a part of the body. The blue coloring of the goblet cells tells us that mucous proteins do not carry the same ionic charges as most proteins found within the epithelial cells of the pseudo-stratified epithelium. Due to this difference in color, they are classified as a distinct cell– a unicellular gland found within the pseudo-stratified epithelium. But what does our friend the embryologist say? Well, the columnar cells can’t become goblet cells, and the goblet cells can’t become columnar cells, but they do share a common ancestor (or stem cell), so we probably should consider the goblet cells a part of the pseudo-stratified epithelium. Does it matter? No, but questions like this matter later in the book, it’s good to start practicing now.
Connective tissue (CT)

Figure 2.13: Somites of an early stage embryo, from which connective (and muscle) tissue develop
General characteristics of connective tissue
Many textbooks tell you connective tissue connects 2 other tissues. That’s like saying your smart-phone connects you to other people. It is technically true, but glosses over the fact these could be people you know, people you don’t know, or people you don’t want to know. It ignores the fact you could be communicating synchronously with people on the other side of the world, communicating asynchronously over email, accessing information from the near sum-of-all human knowledge (the internet), or reading the sophomoric rant of some disgruntled knuckle-dragger who, years ago, posted a rude comment on a YouTube video recording of a histology class lecture (also the internet). So far, we’ve tried to keep you, the reader, safe from extraneous information about histology that is not be relevant to your career. Here we go into more detail than your average undergraduate histology class.
Connective tissues come from segments of tissue in an embryo, found between two layers of epithelium, called somites (Fig. 2.13). Because of their shared lineage, connective tissues share a number of common features. The average connective tissue has few cells, and is mostly composed of Extra-Cellular Matrix. That matrix includes glycoproteins and polysaccharides made by the cells, which attract water to form the gel matrix of ground substance. It also includes more visible fibers. Some important ground substance molecules covered earlier are Fibronectin and Hyaluronic Acid. The three main types of visible fibers are collagen, reticular and elastic fibers. Connective tissues are typically highly vascular, meaning they contain blood vessels.
There are a number of different cell types found within a connective tissue. The stem cells are mesenchymal stem cells (MSCs). Mesenchymal stem cells are extremely important cells (here is further reading), and since you can find them in adult tissues they are called a type of adult stem cell. These cells are capable of undergoing mitosis to produce more adult stem cells, which can differentiate into fibroblasts, lipoblasts, chondroblasts, osteoblasts, hemocytoblasts, myoblasts and neuroblasts. Thus, they form most connective tissues, including bone, cartilage, blood (both red and white blood cells), plus muscle tissue. That’s pretty much everything except epithelia, which mesenchymal stem cells can form as well after going through a transition.

Figure 2.14: Fibroblasts, as seen under a confocal microscope. Image credit: Indian Muntjac fibroblast cells By Michael W. Davidson is licensed CC BY 2.0
In a basic connective tissue, mesenchymal stem cells divide and differentiate into fibroblasts, the cells that blast out the fibers and ground substance found in ECM. There may be other cells found in a connective tissue, including adipocytes, red and white blood cells, or other cells that have emigrated from their tissue of origin. In a mature connective tissue, the fibroblasts are sometimes called fibrocytes, which fits with our regular nomenclature for a cell going through stages of differentiation. More frequently, however, they are called fibroblasts and we don’t worry about whether they are actively blasting out fibers at the time or taking a rest.

Figure 2.15: Mesenchyme, the generic embryonic connective tissue, doesn’t have any specialization. Image credit: Human Mesenchyme By Jpogi at Wikipedia is in the Public Domain CC0
Mesenchyme
Mesenchyme is the first type of connective tissue an embryo makes. It differentiates into other connective tissues. It is an embryonic tissue that has yet to decide what it will become when it grows up. It is composed mainly of mesenchymal stem cells and mucous ground substance (mucous is an adjective to describe loose ECM, while mucus is a substance excreted from the body). There is a special type of mesenchyme important to face and tooth development, called neuro-mesenchyme. The lineage of neuro-mesenchyme is not from mesoderm like most connective tissues, but from a special type of neuronal cell called a Neural Crest cell. Neuro-mesenchyme forms special connective tissues we have yet to mention: dentin, cementum and the periodontal ligament.
Figure 2.16: Areolar connective tissue, histology and cartoon.
Areolar connective tissue
Areolar connective tissue is the quintessential connective tissue, or the most boring depending on how you look at it. It contains a little of everything other connective tissues might have: cells, ground substance, and all 3 fibers. Because it has a good amount of ground substance, it is an ideal tissue to occupy places where blood vessels might need space to grow in the future. Hence areolar CT is found in regions that are highly vascular. This is why you can find a small layer of areolar connective tissue directly underneath nearly every epithelium, including the stratified squamous epithelium of the various types of oral mucosa. Ground substance also absorbs water and swells in size, which is why the areolar connective tissue of the nasal cavity is also referred to as erectile tissue.
Figure 2.17: Dense irregular connective tissue, histology and cartoon
Dense irregular connective tissue
Underneath the areolar CT of the oral mucosa is dense irregular connective tissue. Fibroblasts make the bulk of the tissue: collagen fibers, the strongest of the 3 fiber types. In a dense irregular CT, the collagen fibers point in all directions. This makes this tissue particularly strong in all directions. In Fig. 2.17, most of the pink is extracellular collagen fibers, and the white space ground substance. You can find dense irregular CT (along with its buddy, areolar CT) in the dermis of the skin. Why in both places, you ask? Lineage.

Figure 2.18: Histology of dense regular connective tissue.
Dense regular connective tissue
Like dense irregular CT, dense regular connective tissue is mostly collagen fibers, only the fibers run parallel to one another. That makes this tissue strong in one direction. You mostly find dense regular CT between muscle and bone, or between bone and bone. The only place in this book where we find dense regular CT is the periodontal ligament, between bone and cementum.
You might be thinking the collagen fibers look wavy, not parallel. When someone sliced this section of a tendon, they sliced through the tissue using a thin blade. As a blade cuts the tissue, the tissue moves, and so the blade pushes fibers in one direction, then pulls them back, and pushes again as the person slices as thin of a slice as they can. For better results, use an expensive device that freezes or embeds soft tissues in wax, such as a microtome, cryotome, or vibratome. We bring this up because many textbooks suggest histology is a nearly-perfect representation of what is found in the human body, when in reality there are artifacts, or errors, that histologists have learned to ignore when we see them. We have chosen to illustrate many of the images in this book to avoid discussion of artifacts where possible, and focus on concepts relevant to dental hygiene.
Figure 2.19: Adipose connective tissue, histology and cartoon
Adipose connective tissue
Adipose tissue is a type of connective tissue. It is special in that is has very little ECM. It contains mostly adipocytes, the mature cells that store triglycerides. It also contains mesenchymal stem cells, which if a person needs more adipose tissue, divide and differentiate into more adipocytes. Technically, Mesenchymal Stem Cells differentiate into a lipoblast first, but this is only important for the sake of staying consistent with the naming system. Maybe if they had named the cells adipoblast and adipocyte, we’d use the -blast name more frequently, but someone messed that up.
Many textbooks place adipose tissue next to areolar connective tissue and categorize them both “loose”, on account of their low number of visible fibers. That’s fine if you like categories based on appearances, but you don’t need to call either of them “loose”. It is descriptive and does not correspond to anything embryological. We find adipose tissue in many places throughout the body, either between two other tissues, or embedded within another tissue (such as the adipose you find in bacon). On the other hand, areolar connective tissue is found next to dense irregular connective tissue, which is why we’ve deviated from the grouping found in most textbooks.

Figure 2.20: Hyaline cartilage, histology and illustration. Order of appearance: lacunae, chondrocytes, ground substance, perichondrium, surrounding dense connective tissue.
Cartilage connective tissue
Cartilage connective tissue comes in three basic flavors: hyaline cartilage, fibrocartilage and elastic cartilage. The hallmark feature of cartilage connective tissue is its ample amount of ground substance. Chondroblasts secrete glycosaminoglycans and glycoproteins, such as Hyaluronic Acid, which attract water, forming a very dense but slippery gel. The ECM of cartilage contains collagen– there is more collagen in the fibrocartilage of the temporo-mandibular joint, whereas the elastic cartilage of the epiglottis has more elastic fibers. After secreting ECM, the chondroblasts differentiate into chondrocytes, and reside within the tissue in spaces called lacunae (lakes). The outer edges of cartilage are a thin layer of parallel collagen fibers known as the perichondrium. If you look at Fig 2.20, the perichondrium appears like a layer of dense regular connective tissue (it is, it just has a second name), and the edge of the perichondrium blends into dense irregular connective tissue. The reason the cartilage blends into dense irregular connective tissue is they share the same lineage: chondroblasts and fibroblasts both differentiate from mesenchymal stem cells.

Figure 2.21: Bone connective tissue, histology and cartoon highlighting osteocytes and lamellae of ECM.
Bone connective tissue
Bone tissue (or osseus tissue) is the last type of connective tissue we cover in detail. Because of its similarity to dentin, cementum and enamel, we cover bone tissue in a fair amount of detail. Like any connective tissue, bone tissue starts as mesenchymal stem cells, which differentiate into an intermediate stem cell called an osteo-chondro-progenitor cell, which can in turn decide to adopt an osteoblast or a chondroblast fate, depending on what signals it receives. A stem cell that has adopted the osteoblast fate (and denied the chondroblast fate) would be an osteo-progenitor cell. These osteo-progenitor cells are stem cells within the connective tissues surrounding bones, the periosteum (superficial) and endosteum (deep). When stimulated, they undergo mitosis, and one daughter differentiates into an osteoblast. An osteoblast is the cell that secretes the ECM that makes up the bulk of bone tissue. Osteoblasts secrete collagen fibers and ground substance, which later mineralizes, trapping the osteoblasts inside of a lacuna (a small space within dense ECM). At this point, they differentiate into osteocytes and maintain the bone tissue. Next, a new set of osteoblasts lay down another layer of bone tissue around the previous one. This creates either the concentric layers of bone tissue found in the osteons of compact bone, or the thinner spiral layers of bone tissue found in the trabeculae of spongy bone.
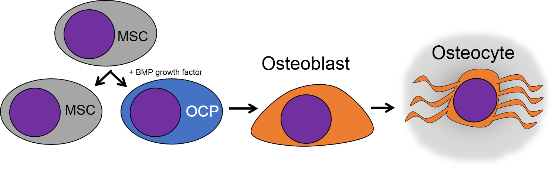
Figure 2.22: Three (of four) major cell types found in bone tissue and their lineage. MSC = mesenchymal stem cell, OCP = osteo-chondro progenitor cell.

Figure 2.23: Calcium hydroxyapatite crystal structure of bone ECM. Legend: Red/orange = phosphate, white = calcium ions, yellow = hydroxide ions. Image credit: Fluorapatite by Von Benjah-bmm27 in the Public Domain CC0
The extra-cellular matrix of bone tissue is roughly 1/3 collagen fibers (organic protein) and 2/3 mineral (inorganic). The mineral component is a mixture of positive-charged Ca2+ ions, which react with negative-charged phosphate (PO43-) to form a hard, crystalline matrix. Because most cells are full of phosphate, Ca2+ levels in the cytoplasm must always be kept very low, so if a cell like an osteoblast wishes to store Ca2+, it does so within its smooth Endoplasmic Reticulum. The Ca2+PO43- further react with water (or more specifically, hydroxide ions OH–) and small amounts of fluoride(F–) to form a crystal named calcium hydroxyapatite. Dentin, enamel and cementum have ECM very similar to bone tissue, only with varying amounts of collagen versus mineral components.
Collagen fibers run parallel within one layer of bone tissue, and 90° in the next layer. While the mineral component of bone and tooth tissues provides strength, the collagen provides the ability to bend or flex, reducing the chances the tissue will shear off under stress. Collagen fibers, therefore, have a function similar to the rebar in reinforced concrete. The lower percentages of collagen found in enamel makes it harder and more resistant to caries than dentin or cementum, but still susceptible to fracture if enough force is applied.

Figure 2.24: Collagen fibers of a tendon are continuous with the periosteum and Sharpey’s fibers of bone tissue.
Bones are surrounded by a layer of dense regular connective tissue called the periosteum. This tissue is a continuation (not a separate structure) of the same tissue of tendons and ligaments. The periosteum also contains osteo-progenitor cells, the semi-differentiated cells that came from Mesenchymal Stem Cells, as well as osteoblasts and osteoclasts (coming up). Collagen fiber bundles from this layer named Sharpey’s fibers penetrate the superficial layers of compact bone. This makes a very strong connection between bone tissue and tendons or ligaments.

Figure 2.25: Osteoclasts demineralize bone tissue.
Another important cell found in the periosteum and endosteum is the osteoclast. This cell is not derived from the osteo-chondro-progenitor cell that gives rise to osteoblasts and osteocytes. The lineage of an osteoclast traces to the bone marrow, from a stem cell that also gives rise to red and white blood cells. An osteoclast is a close relative of a white blood cell called a macrophage, more than it is to bone cells. Osteoclasts demineralize bone tissue, releasing Ca2+ into the bloodstream. This is important because muscle and nervous tissue cannot function without Ca2+, which our diet cannot provide continuously. Hence, bone tissue can be thought of as a calcium storage organ.

Figure 2.26: The bone remodeling unit: osteoblasts plus osteoclasts. Image credit: Own work By Shandristhe Azylean is licensed under CC BY-SA 3.0
Osteoclast activity is important during the exfoliation of teeth, as bone tissue is removed to loosen the connection between bone and tooth. Osteoclasts activity is also key to the mechanism by which orthodontia works. Lastly, osteoclasts play an important role in maintaining bone health– this may seem counter-intuitive, because osteoclasts destroy bone tissue. Bone tissue is constantly being repaired by a group of osteoblasts and osteoclasts working together, known as a remodeling unit. Because compact bone tissue is dense, there is little room for cells to work. Osteocytes can repair small amounts of damage, but larger amounts of damage would build up over time. To prevent this, remodeling units constantly work to remove bone tissue and replace it with fresh bone tissue. The remodeling units cannot find damaged bone tissue, they simply keep removing and replacing bone tissue. Physical stress on bone tissue causes osteoblasts to work a bit harder, which leads to increased bone density. Loss of osteoblast-stimulating hormones (such as estrogen) can cause osteoblasts to work more slowly than osteoclasts, which can lower bone density and potentially lead to osteoporosis.

Figure 2.27: The basic steps of endochondral ossification.
Bones form either by endochondral ossification, or by intra-membranous ossification. The two processes are similar, the major difference is that endochondral ossification begins with a hyaline cartilage model, whereas intra-membranous ossification begins with a dense connective tissue model. Most of the skull develops by intra-membranous ossification, except the mandible and maxilla, which use both. Because of their similarity, we cover only endochondral ossification.

Figure 2.28: Blood vessels and nerves develop first, and are later surrounded by bone tissue. Image credit: “Dissected skull, Maison Tramond model, Paris, nineteenth century” by Centre for Research Collections University of Edinburgh is licensed under CC BY-SA 2.0
Most of the skeleton starts off not as bones, but as cartilage. Cartilage is typically avascular, but when chondrocytes receive the correct signals, blood vessels grow into the ground substance. The growth of new blood vessels into a tissue is called angiogenesis. Angiogenesis provides the pathway that mesenchymal stem cells use to migrate into the central area, where they differentiate into osteoblasts. After that, osteoblasts replace cartilage tissue with spongy bone tissue. This first site of ossification is named the primary ossification center. As remodeling occurs at the primary ossification center, blood vessels grow into the epiphyses and begin the same process at what are named the secondary ossification centers. Ultimately, most of the cartilage is replaced, except in two important places. First, cartilage may remain between the primary and secondary ossification centers, leaving growth plates (such as in bones of the arms and legs). Cartilage also remains at the ends, providing articular cartilage found in synovial joints, such as the temporo-mandibular joint. Intra-membranous ossification, because it begins with a dense connective tissue, leaves fontanels (soft-spots) between bones until ossification is complete, rather than cartilaginous connections.

Figure 2.29: The basic steps of bone repair. Note the similarity to endochondral ossification.
When bone tissue is injured, it goes through a healing process similar to endochondral ossification. Perhaps you remember mention of wound healing recapitulating development? Here is an example of an adult tissue re-using the same mechanism to heal that it used during embryogenesis to grow in the first place. Similar to an injury to the skin, bone tissue first goes through an inflammatory process, and damaged blood vessels form clots. External blood clots are known as scabs, but inside the body they are named a hematoma. A blood clot contains an ECM fiber named fibrin that acts as a scaffold. This scaffold allows blood vessels to grow into the area, by angiogenesis. This in turn allows mesenchymal stem cells to migrate into the injured area, leave the blood and migrate along the scaffold, and differentiate into chondrocytes. The chondrocytes replace the fibrous hematoma with fibrocartilage– this cartilage step is really the only step that is does not have a homologous step (a shared step) in wound repair in the skin. Unlike repair of the skin, bone tissue growth needs a cartilage model first. With this new cartilage scaffold, more mesenchymal stem cells migrate and differentiate into osteoblasts, and replace the cartilage with bone tissue. Later, spongy bone tissue is remodeled so that it roughly matches the original compact and spongy layers. One difference can be seen afterwards, the former site of injury is a little bit thicker after it is repaired—similar to the way skin tissue can be made stronger after an injury because the collagen fibers in scar tissue have more covalent cross-links than in dense irregular CT.

Figure 2.30: Artificial scaffolds help mesenchymal stem cells migrate into an injured area, differentiate into osteoblasts, and produce bone tissue. Image credit: Alloplastic particulate graft by Coronation Dental Specialty Group is licensed CC BY SA 3.0
Knowing the steps of endochondral ossification and bone fracture repair are important in instances where stimulating or mimicking the natural healing process can reduce the need for surgeries and implants. For instance, patients may lose a significant amount of bone tissue with prolonged periodontitis. If enough bone tissue is lost, a surgeon may use a bone tissue graft to replace it. Bone tissue from a cadaver can be ground up, destroying the cells but leaving the collagen and minerals. Other sources may include a patient’s own bone tissue (a full set of ribs or fibulas is less important than an intact set of jaw bones and teeth). The harvested ECM is spread into the injured site., where it acts as a scaffold, not as a replacement. The patient’s mesenchymal stem cells migrate through the scaffold and begin the healing process, which can proceed quickly because all the raw materials are in close proximity. The grafted scaffolding does not have to come from actual bone tissue. It can be ground-up dentin, fibrin, or synthetic polymers printed using a 3-D printer. The scaffolds, synthetic or biological, are ultimately replaced, which is why you may see this referred to as Guided or Resorbable Tissue Regeneration. Later we learn a scaffold is just one part of the healing process, there are important signals that can be used to guide mesenchymal stem cell migration, division and differentiation. If you wish to know more, We have a short YouTube video that covers this new technology in more detail, aimed at 200-level Anatomy and Physiology students.

Figure 2.31: Mineralized tissues first evolved in the ectoderm (similar to enamel and were later produced by mesoderm (such as bone). Image credit: “The origin of bone. “ by Darja Obradovic Wagner and Per Aspenberg is licensed under CC BY-NC-SA 4.0
Lastly, it is interesting to consider, from an evolutionary viewpoint, the lineage of the four mineralized tissues (bone, enamel, dentin and cementum) in humans and other vertebrates. Did one evolve first, and the others modified from the parent tissue? Fig. 2.31 provides a hint. We consider this question of lineage from a developmental viewpoint, in chapters 8-11.
![[cut output image]](https://i0.wp.com/histology-and-embryology.com/wp-content/uploads/2020/08/cut-output-image.gif?resize=255%2C296&ssl=1)
Figure 2.32: Hypothetical comparison between blood connective tissue and reticular connective tissue
Blood, Reticular, and Lymph Connective Tissue
There are other connective tissues covered in histology classes, here we cover them briefly. Blood is a type of connective tissue, composed of cells (red blood cells, mostly), liquid ground substance (blood plasma) and fibers (fibrin). Lymph is primarily white blood cells, plus liquid ground substance. Reticular connective tissue contains blood cells, liquid ground substance and reticular fibers. It can be found in lymphatic organs where blood cells tend to rest rather than flow, such as lymph nodes.

Figure 2.33: Scar tissue in the heart after a myocardial infarct. Image credit: “Histopathology of dense fibrous scar replacing myocyte loss in myocardial infarction” by Katarzyna Michaud et al is licensed under CC BY-SA 4.0
There are other forms of connective tissue that aren’t typically covered in 200-level histology classes, we cover one of them: scar tissue. Scar tissue is made by fibroblasts following an injury. This occurs after an injury to any tissue that is large enough that it cannot be replaced quickly by stem cells. One example is sclerotic dentin. Scar tissue is very similar to dense regular connective tissue, except the collagen fibers in scar tissue are highly cross-linked. This makes scar tissue very strong, but also reduces its mobility and capacity to be remodeled. Scar tissue can be formed wherever there are large injuries to an organ– fibroblasts can often fill up the space more quickly than waiting for regeneration to occur. Once formed, scar tissue is difficult to remodel, so it is more-or-less permanent.
Muscle tissue

Figure 2.34: Somites, from which connective tissue and muscle tissue develop.
General characteristics of muscle tissue
Muscle tissue, like connective tissue, is derived from somites. The stem cell that differentiates into one of the several types of muscle cells is called a myo-satellite cell. Given the correct set of extra-cellular signals, myo-satellite cells differentiate into skeletal, smooth or cardiac muscle cells. Skeletal muscle cells are long multi-nucleate cells that were once hundreds of myoblasts that fuse together. Like a fibroblast, a myoblast is stem cell that has undergone some differentiation, enough to limit its fate, and is responsible for building the adult tissue. In muscle tissue, myoblasts create protein fibers that are mostly intra-cellular (such as actin thin-filaments and myosin thick filaments). After fusing with other myoblasts, the adult muscle cells are often confusingly referred to as muscle fibers, but they are definitely cells, not molecules like the protein and glycoprotein fibers found within the ECM of connective tissue. Thus, they can also be called myocytes. In fact, they aren’t the only multi-nucleate cell you read about in this chapter, is perplexing as to why this cell often receives a special suffix, while osteoclast cells do not. In adult muscle tissue, myo-satellite cells remain, ready to undergo mitosis and fuse to form larger muscle cells in response to exercise or injury. We see skeletal muscle in the tongue and underneath regions of oral mucosa, but we do not cover smooth or cardiac muscle tissue.

Figure 2.35: Muscles (organs) contain muscle tissue and dense regular connective tissue.
Muscle tissue is composed primarily of muscle cells, connected by collagen fibers. Unlike connective tissue cells, which may bind to collagen or other ECM proteins for migration using trans-membrane linkers such as integrins, muscle cells anchor in place to the ECM. Muscle cells express different linker proteins to do this, such as the protein Dystrophin. The layers of collagen are thin between cells, and thicker on the outer surface of a muscle. These layers of collagen fibers ultimately become the dense regular connective tissue of a tendon. It is often difficult to determine where muscle tissue ends and connective tissue begins, because of the collagen fibers in both tissues. By extension, it is also difficult to tell where the tendon ends and the periosteum begins. Some histologists draw lines between these tissues, but the junction is better described as a blending. This is because both muscle tissue and connective tissue have the same lineage, from the mesenchyme of somites.
For people studying musculo-skeletal diseases like Muscular Dystrophy, there is interest in identifying the instructions that guide mesenchymal stem cells to differentiate into myo-satellite cells– especially considering the larger number of mesenchymal stem cells in the bone marrow compared to myo-satellite cells in the muscle of a child or adult. If novel treatments result from this research, it could make dental hygiene procedures for patients with Muscular Dystrophy much simpler.
Nervous tissue

Figure 2.36: Confocal image of neural tissue, including neurons and glial cells. Image credit: Desert rose by Tarzerind is licensed under CC By SA 4.0
General characteristics of neural tissue
Shown to the left is a confocal image of neural tissue. What I like about this image, and is often absent from images of neural tissue, is that this one shows all of the cells. The typical image of neural tissue uses stains to highlight neurons—the cells specialized at conducting electricity– leaving out the glia—the majority of cells in the nervous system that do everything else, including guide neurons connections and change the strength of those connections when we learn. What you should see at this time is neural tissue is composed primarily of cells, and has very little ECM. While the brain especially has a huge number of blood vessels inside of it, technically they do not run within the neural tissue. Instead, there is a thin blood-brain-barrier separating the brain from the blood vessels. Normally, we wouldn’t call neural tissue avascular, we do so now only to make a point: neural tissue shares a lot in common with epithelial tissue. By now, we hope you have guessed why: neural and epithelial tissues share the same lineage.
3 major embryonic tissues
Figure 2.37: Animated illustration of gastrulation.
Gastrulation
When an embryo implants in the uterus, it is a hollow ball of identical, omnipotent stem cells that resemble a simple cuboidal epithelium. But soon, some of these epithelial cells migrate inwards, in a very important process called gastrulation. At this stage, we have two distinct layers of cells. The layer on the outside becomes the epidermis, oral mucosa and the central nervous system. The layer one on the inside becomes the lining the gut and other hollow organs. Next, some cells from the outer layer change. They lose contacts with neighboring cells, and migrate into the middle. As this happens, they stop looking like epithelial cells and begin to look like mesenchymal cells. This produces a third layer of cells in an embryo. The three layers are listed below, along with what they form in the adult:
| 3 embryonic layers | Cell fate |
|---|---|
| Ectoderm |
Epithelium of skin and oral mucosa, neural tissue |
| Mesoderm |
Connective and muscle tissue |
| Endoderm |
Epithelial lining of hollow organs |
Table 2.3: the three embryonic tissues
We have now sneakily introduced you to two important terms used in development. The first is cell lineage, which means where a cell came from in the past, and now cell fate, which means what that cell can become in the future. If we didn’t think your license exam would include questions on the four major tissue types, we would stick with the three embryonic tissues, with neural being a sub-family of epithelia (you’ll see why later). We’d have put connective and muscle tissue within the same lineage, rather than separate them. But we can see why academia hasn’t changed the way it teaches histology, the confusion that comes with change would not be worth the benefits we’d gain by grouping cells by linage rather than appearance. But this, we think, is more than enough basic histology for a dental hygienist, it is time to move on to the histology of the oral cavity.






